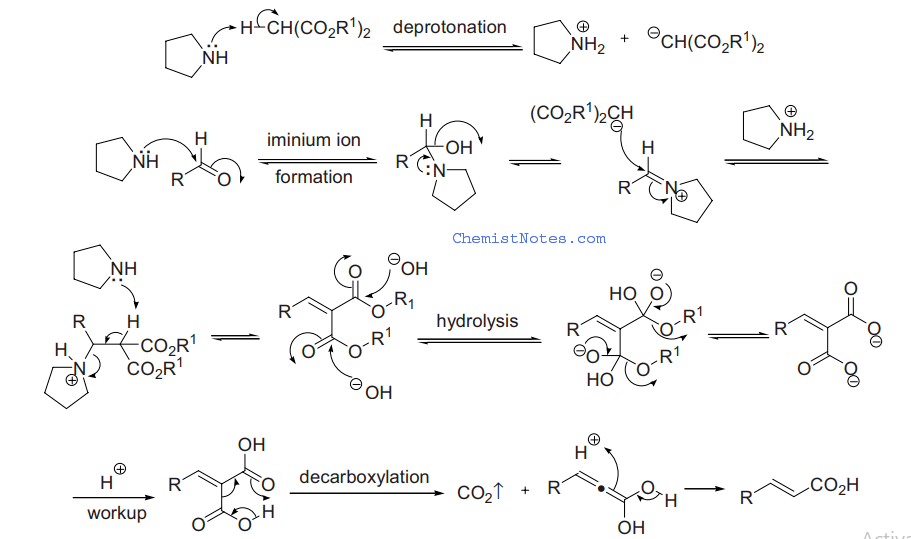
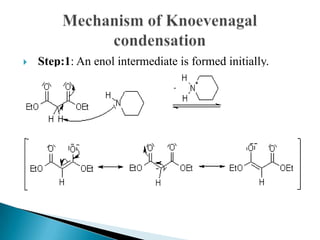
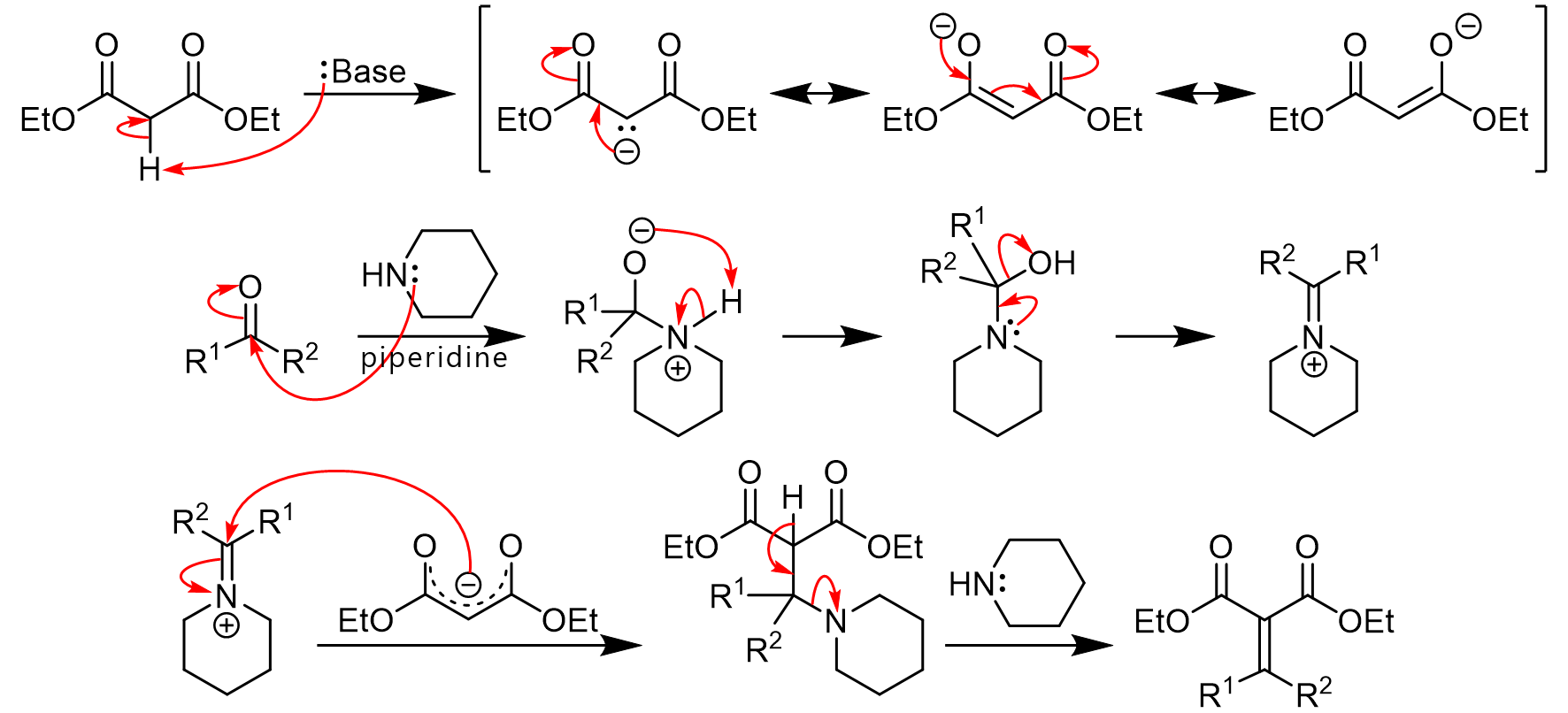
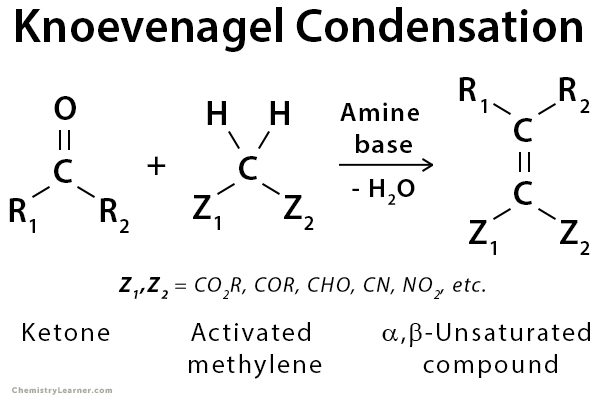
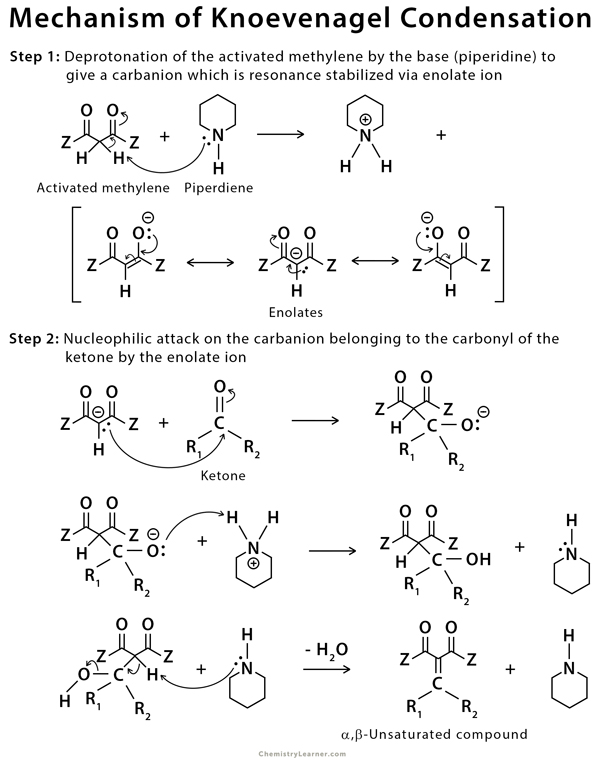
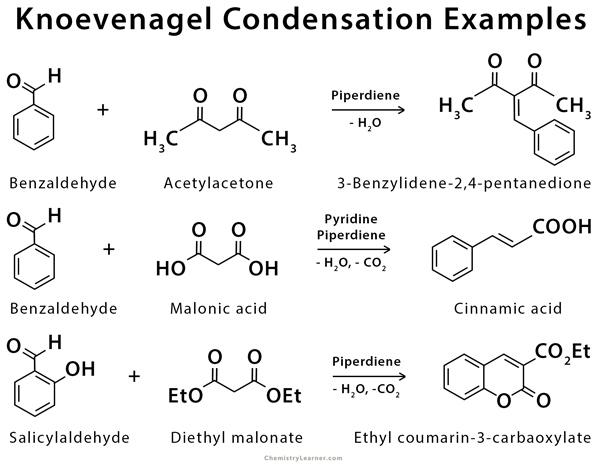
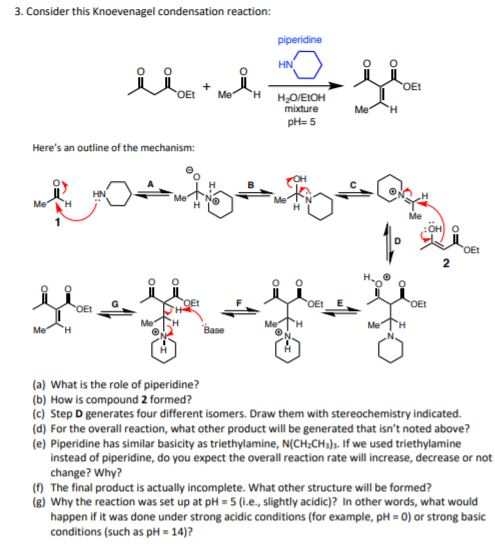
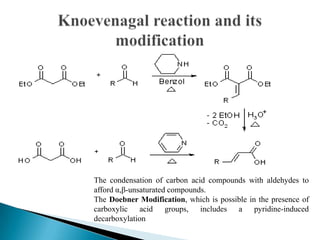
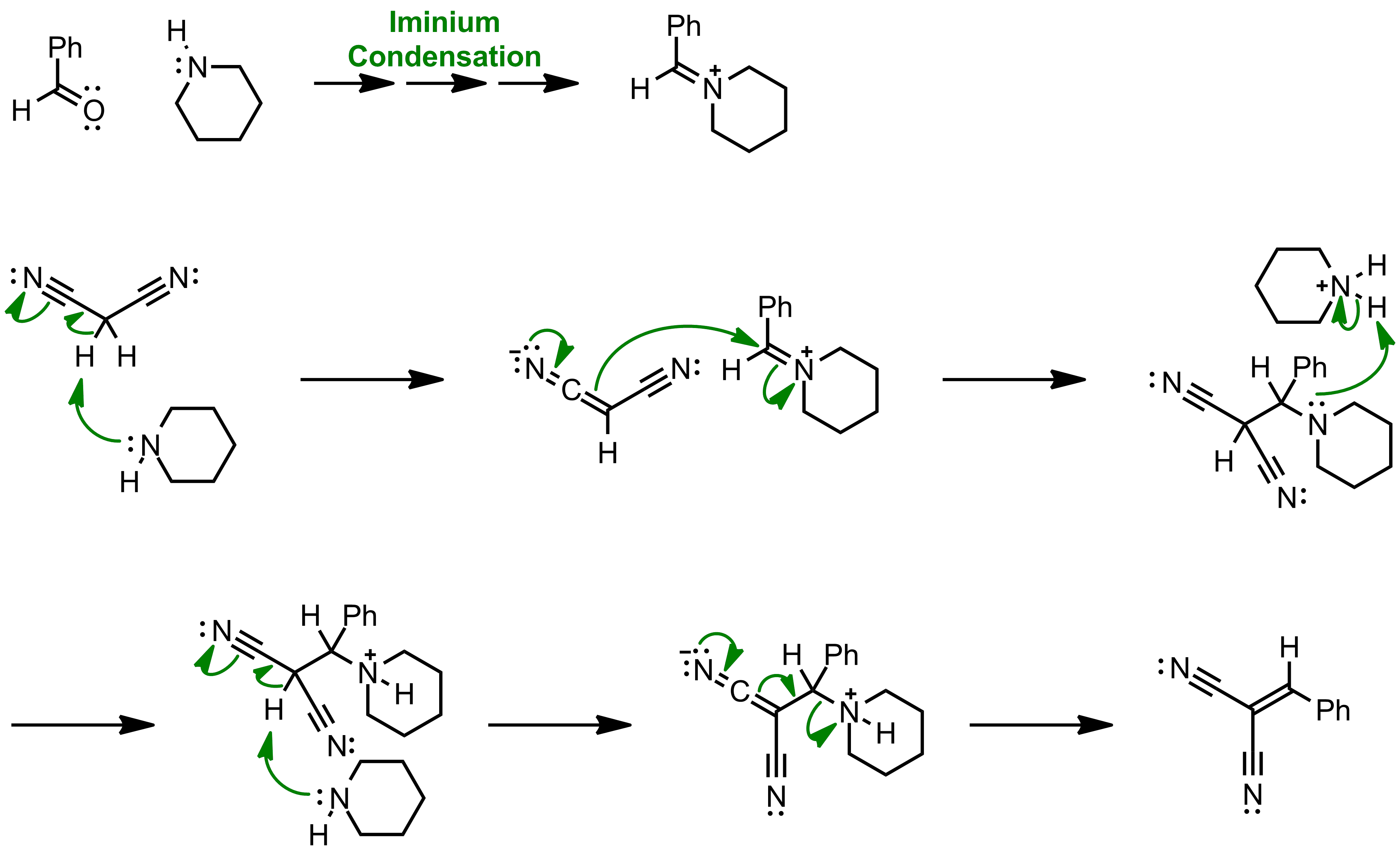
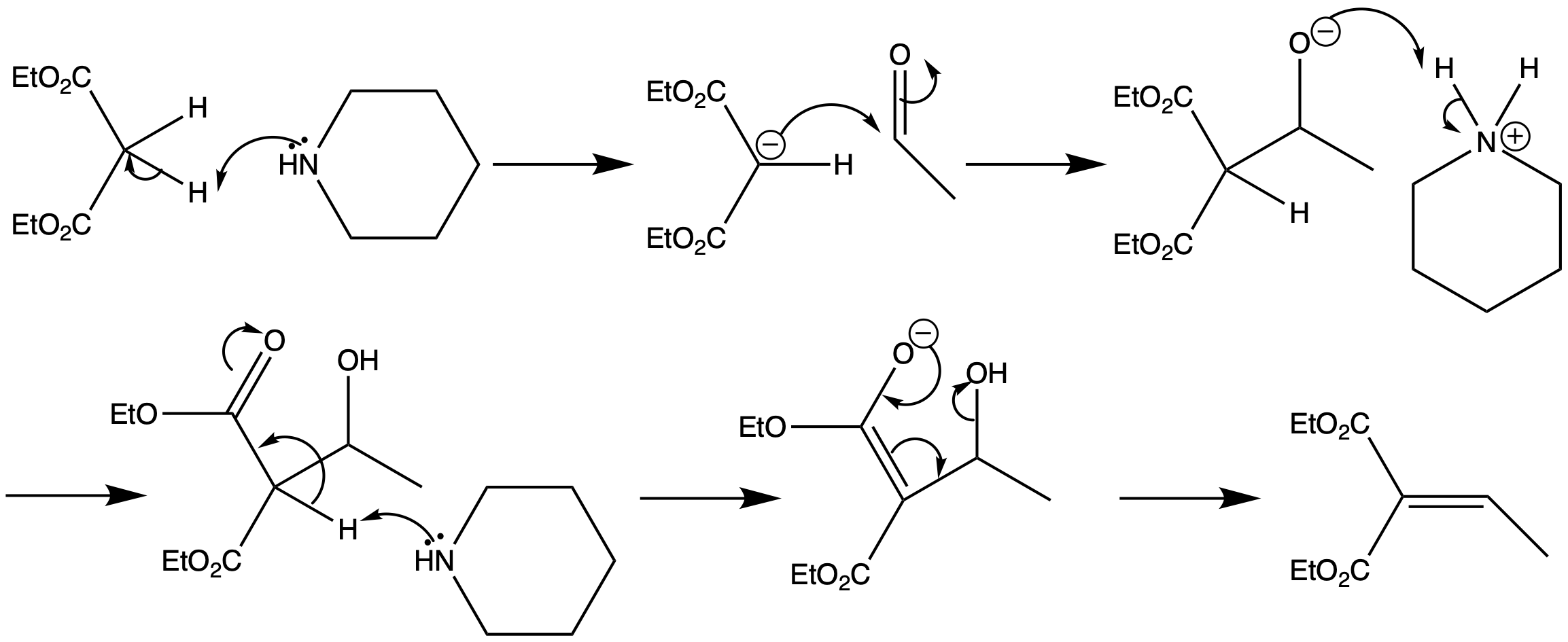
The Knoevenagel reaction is a carbonyl condensation reaction of an ester with an aldehyde or ketone to yield an \alpha , \beta -unsaturated product. Show the mechanism for the Knoevenagel reaction
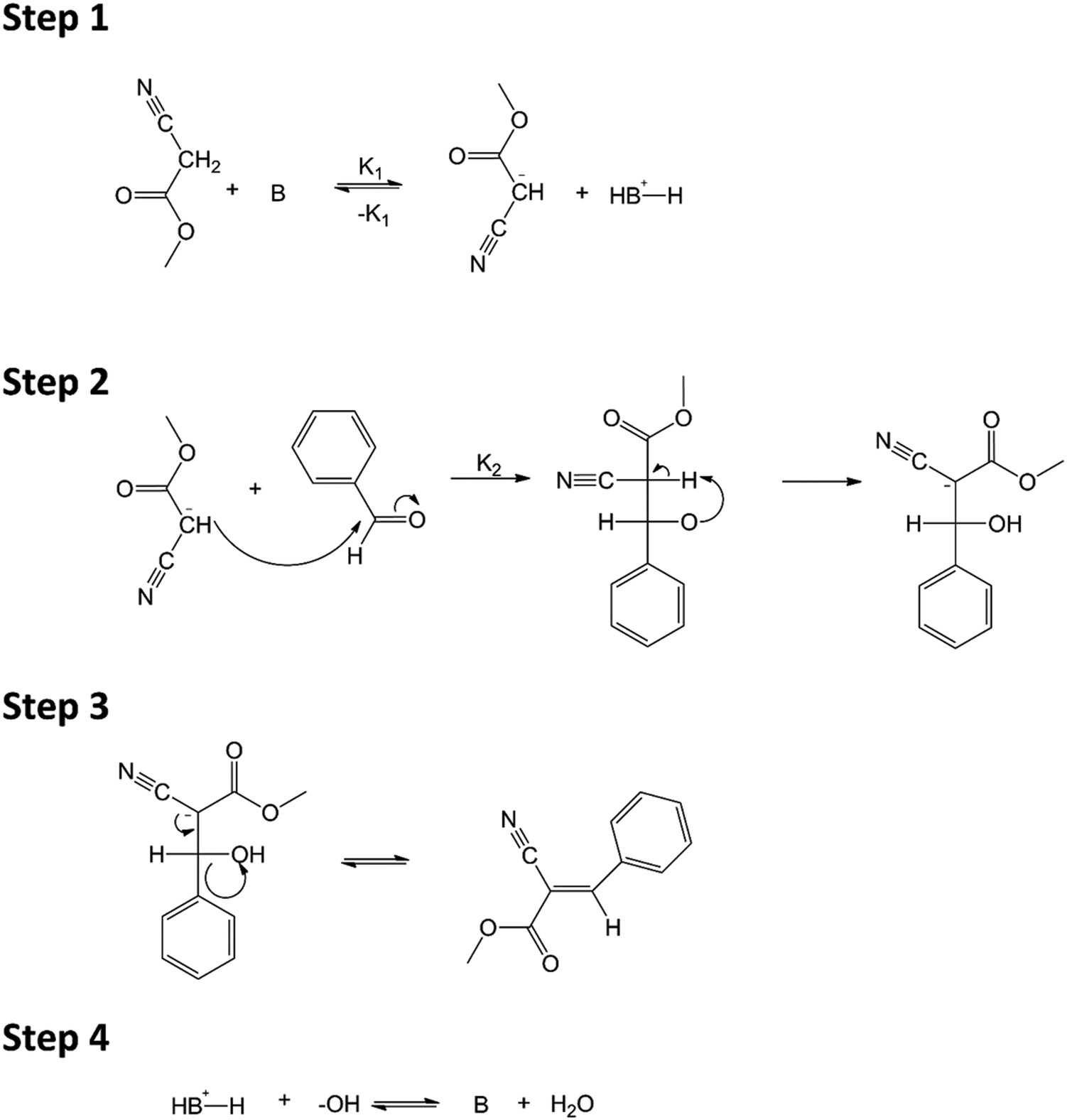
A mechanistic study of the Knoevenagel condensation reaction: new insights into the influence of acid and base properties of mixed metal oxide catalys ... - Physical Chemistry Chemical Physics (RSC Publishing) DOI:10.1039/C7CP04743F

Figure 2 from Mechanism of the Piperidine-Catalyzed Knoevenagel Condensation Reaction in Methanol: The Role of Iminium and Enolate Ions. | Semantic Scholar

Combination of Knoevenagel Polycondensation and Water‐Assisted Dynamic Michael‐Addition‐Elimination for the Synthesis of Vinylene‐Linked 2D Covalent Organic Frameworks - Xu - 2022 - Angewandte Chemie International Edition - Wiley Online Library
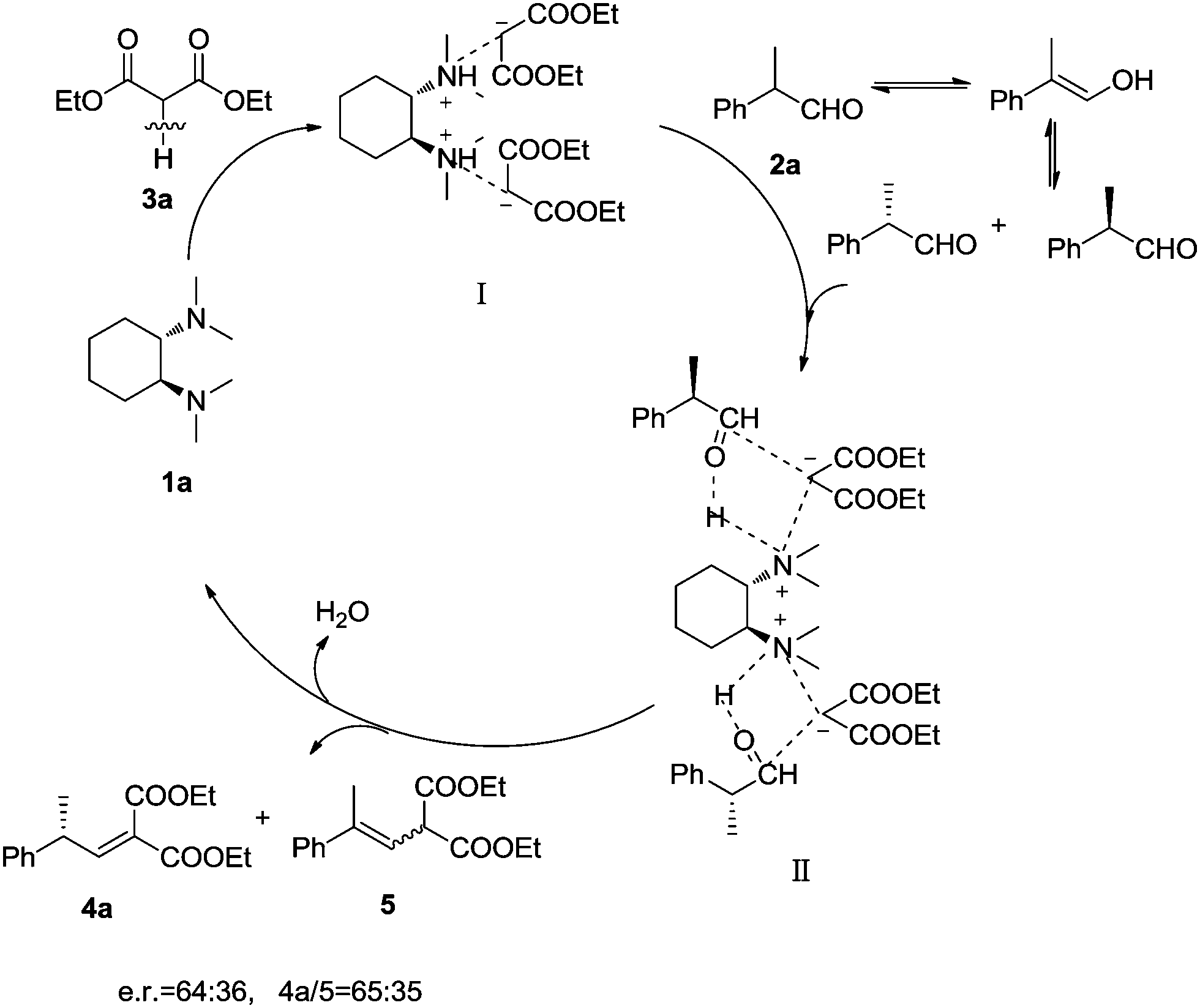
Organocatalytic Knoevenagel condensation by chiral C 2 -symmetric tertiary diamines - New Journal of Chemistry (RSC Publishing) DOI:10.1039/C6NJ00613B